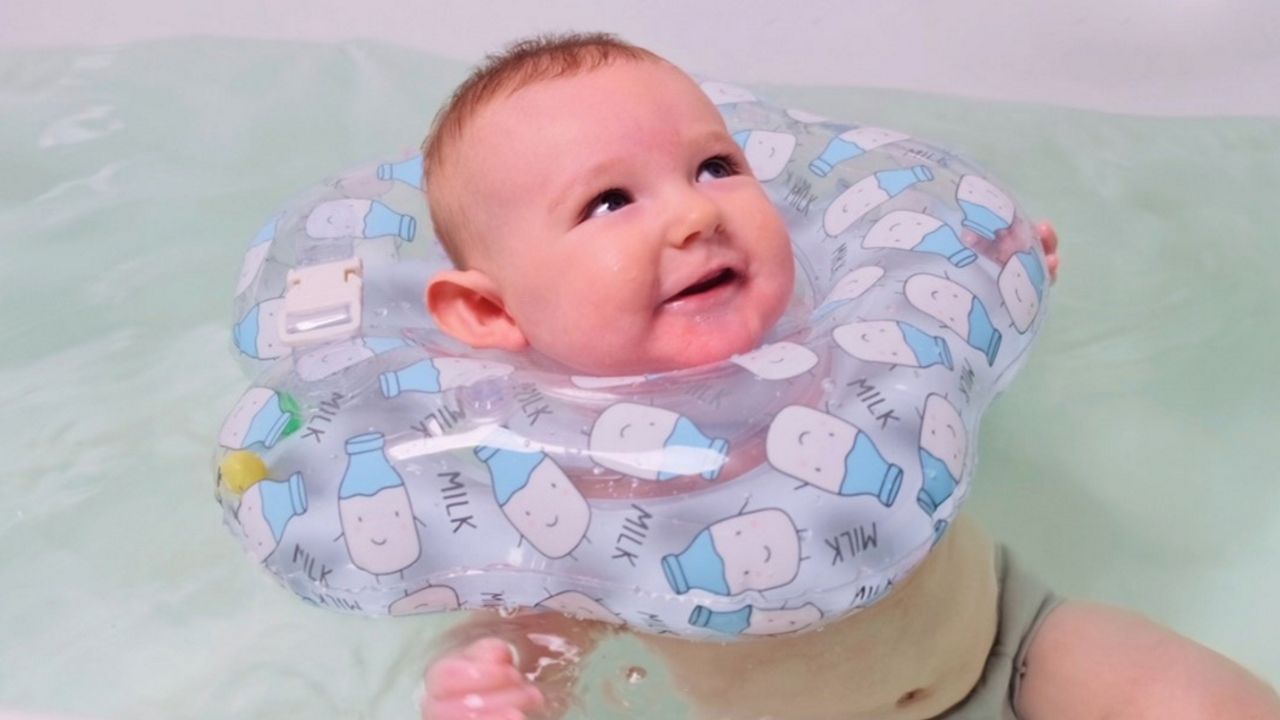
The Food and Drug Administration on Tuesday issued a warning to parents and caregivers across the country, advising them not to use neck floats for infants due to a risk of death or serious injury.
The FDA also warned infants with developmental delays or special needs are at particular risk of suffering an injury or dying due to neck float use, including those with spina bifida, spinal muscular atrophy type 1, Down syndrome and cerebral palsy.
Officials said while such injuries are rare, at least one baby died and another was injured after using a neck float, though there may be other, as-yet-unreported cases. Both cases occurred when the caregiver was not watching the child, the FDA noted.
Some companies have marketed neck floats – which are typically inflatable plastic rings that can be fastened or attached around a baby’s neck – for infants as young as two weeks old and for premature babies. A number of manufacturers claimed the devices promoted health benefits when used in aquatic therapies, including better muscle tone, increased flexibility and range of motion, more lung capacity, better sleep and increased brain function, among others.
“The safety and effectiveness of neck floats to build strength, to promote motor development or as a physical therapy tool, have not been established,” the FDA wrote in part.
The federal health agency is aware of several companies that marketed infant neck floats as a water therapy tool despite not being cleared by the FDA, and has “communicated our concerns about these promotional materials to these companies and will continue to monitor promotional materials and claims for these devices.”
Aquatic therapy has been proven to aid functional mobility for infants and toddlers when used as an early intervention tool – but do not necessarily require the use of a neck float. Preliminary research conducted specifically on the Otteroo, one particular brand of neck float, found the device had “potential for use in aquatic therapy sessions, as part of a home exercise program, or as an adjunct in addition to therapy services,” but the study was too limited to be definitive.
In a statement provided to Spectrum News, Otteroo said it is in the process of "appealing the FDA’s claims that have been put forth against the neck float category in the article."
The company said, while it cannot speak for other manufacturers of baby neck floats, that their product has been endorsed by a number of reputable therapists and organizations, including CureSMA, a non-profit focused on spinal muscular atrophy.
"We’re disappointed that the FDA has not been transparent on why they believe neck strain is a substantial risk," the company added.
The FDA’s Tuesday warning is not the first time health experts have warned of the potential for injuries. Kyran Quinlan, former chair of the American Academy of Pediatrics Council of Injury, Violence and Poison Prevention, in 2019 called neck floats “potential death traps.”
“Neck floats for babies scare me to death, and I hope they scare parents,” Quinlan told Good Housekeeping at the time. “These are potential death traps [...] to have your precious baby one poorly sealed seam away from going under at the pool is frightening.”
This article has been updated to include a statement from Otteroo.







_crop)