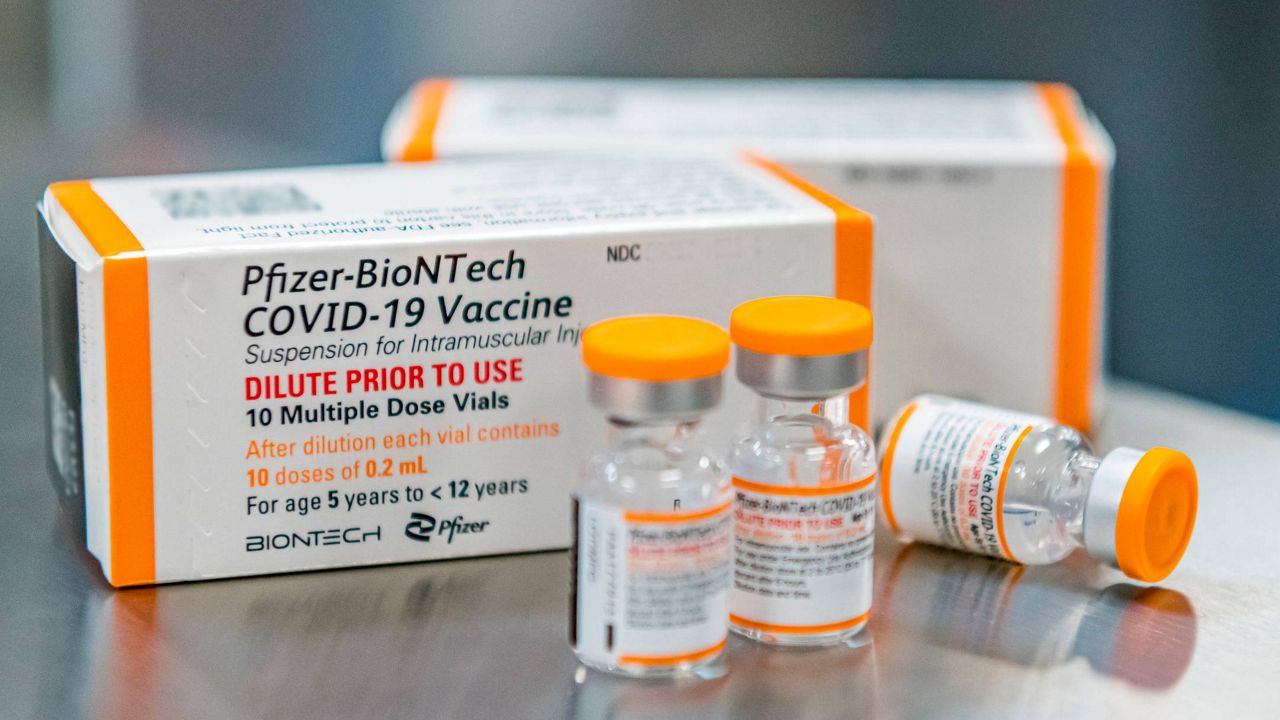Drugmakers Pfizer and BioNTech announced Monday that their COVID-19 vaccine is safe and generated "robust neutralizing antibody responses" in children ages 5-11 in a Phase 2/3 trial, a major step toward authorization for that key age group.
The trial data has not yet been peer reviewed, but the companies plan to "submit data from the full Phase 3 trial for scientific peer-reviewed publication."
Currently, in the United States, the two-dose mRNA vaccine is available for people ages 12 and older. But with kids now back in school and a huge jump in pediatric infections driven by the highly contagious delta variant, many parents are anxiously awaiting vaccinations for their younger children.
The companies tested a much lower dose in children – a third of what is given to adults, administered 21 days apart – but Pfizer said that the vaccine generates antibody levels just as strong as in teenagers and young adults after the second dose.
Side effects – such as fever, achiness or sore arms – were similar to those observed in people ages 16-25.
"Further, the COVID-19 vaccine was well tolerated, with side effects generally comparable to those observed in participants 16 to 25 years of age," the companies added.
"I think we really hit the sweet spot," Dr. Bill Gruber, a pediatrician and a senior vice president with Pfizer, told The Associated Press.
Pfizer and BioNTech said that they plan to submit for emergency use authorization from the Food and Drug Administration (FDA) and other regulatory agencies worldwide "as soon as possible." Dr. Gruber said the companies aim to apply to the FDA by the end of the month for emergency use in this age group, followed shortly afterward with applications to European and British regulators.
“Over the past nine months, hundreds of millions of people ages 12 and older from around the world have received our COVID-19 vaccine. We are eager to extend the protection afforded by the vaccine to this younger population, subject to regulatory authorization, especially as we track the spread of the Delta variant and the substantial threat it poses to children,” Albert Bourla, Pfizer's Chairman and Chief Executive Officer, said in a statement. “Since July, pediatric cases of COVID-19 have risen by about 240 percent in the U.S. – underscoring the public health need for vaccination."
"These trial results provide a strong foundation for seeking authorization of our vaccine for children 5 to 11 years old, and we plan to submit them to the FDA and other regulators with urgency," Bourla added.
“We are pleased to be able to submit data to regulatory authorities for this group of school-aged children before the start of the winter season,” Dr. Ugur Sahin, CEO and co-founder of BioNTech, wrote in a statement. “The safety profile and immunogenicity data in children aged 5 to 11 years vaccinated at a lower dose are consistent with those we have observed with our vaccine in other older populations at a higher dose.”
While kids are at lower risk of severe illness or death than older people, more than 5 million children in the U.S. have tested positive for COVID-19 since the pandemic began and at least 460 have died, according to the American Academy of Pediatrics. Cases in children have risen dramatically as the delta variant swept through the country.
“I feel a great sense of urgency” in making the vaccine available to children under 12, Gruber said. “There’s pent-up demand for parents to be able to have their children returned to a normal life.”
Pfizer said it studied the lower dose in 2,268 kindergartners and elementary school-aged kids. The FDA required what is called an immune “bridging” study: evidence that the younger children developed antibody levels already proven to be protective in teens and adults. That’s what Pfizer reported Monday in a press release, not a scientific publication. The study still is ongoing, and there haven’t yet been enough COVID-19 cases to compare rates between the vaccinated and those given a placebo — something that might offer additional evidence.
The study isn’t large enough to detect any extremely rare side effects, such as the heart inflammation that sometimes occurs after the second dose, mostly in young men. The FDA’s Marks said the pediatric studies should be large enough to rule out any higher risk to young children. Pfizer’s Gruber said once the vaccine is authorized for younger children, they’ll be carefully monitored for rare risks just like everyone else.
Vaccine maker Moderna is also studying its shots in elementary school-aged children. Pfizer and Moderna are studying even younger kids as well, as young as six months. Results are expected later in the year.
The Associated Press contributed to this report.