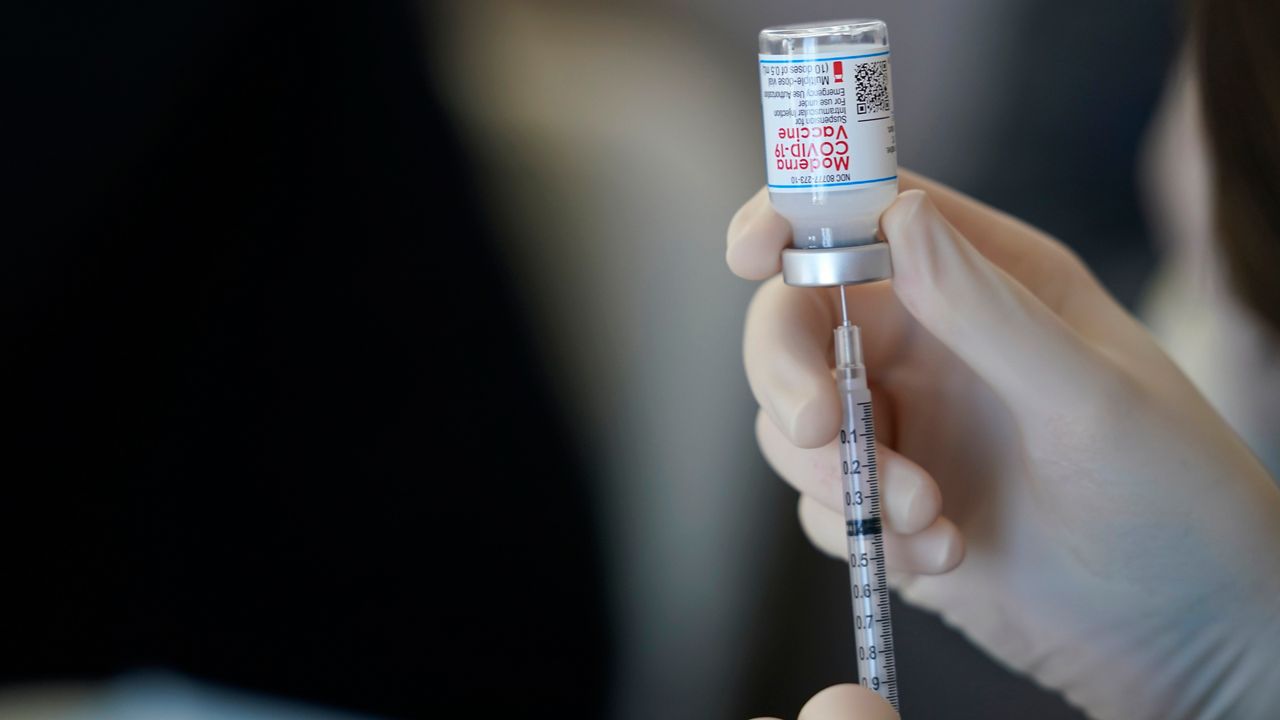
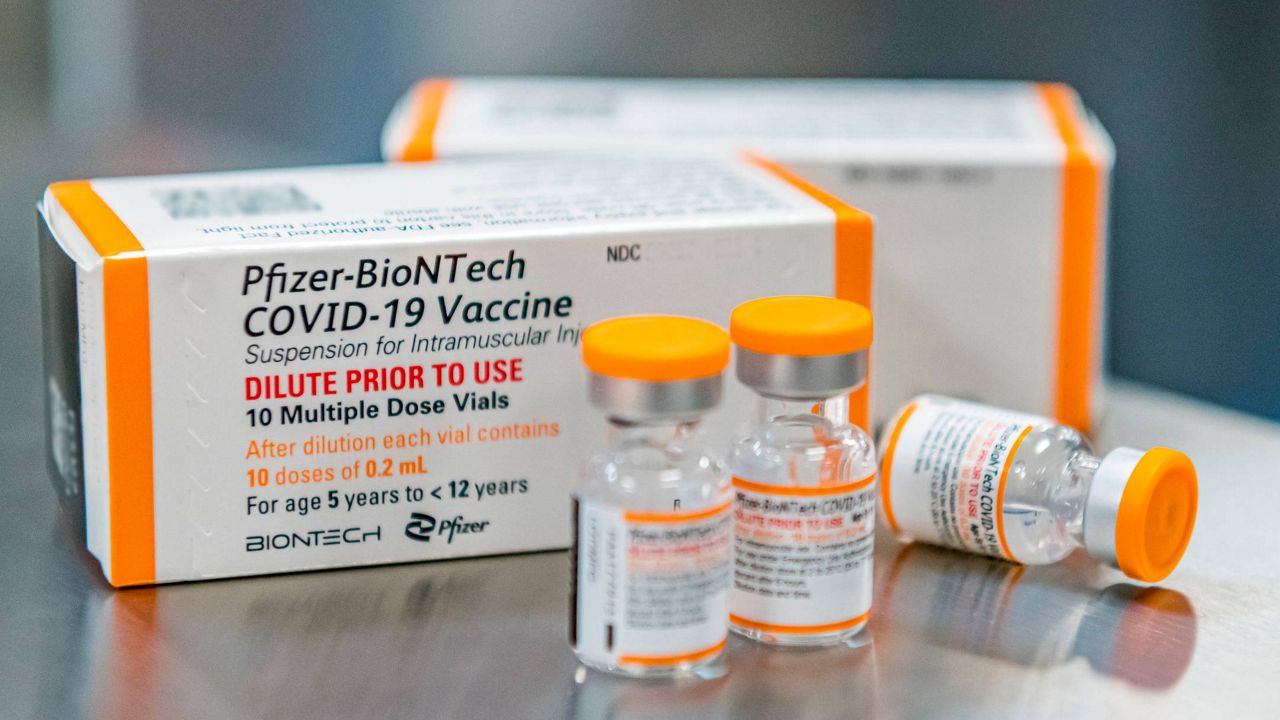Moderna announced Thursday that it has requested emergency use authorization from the Food and Drug Administration for its COVID-19 vaccine for 12- to 17-year-olds.
The Massachusetts-based drugmaker said a clinical trial of more than 3,700 adolescents found the vaccine to be 100% effective at protecting against COVID-19.
Moderna also said there were no significant safety concerns identified in the trial. The majority of side effects were mild or moderate and in line with what adults have experienced — injection site pain, headache, fatigue, muscle pain and chills.
Moderna received FDA emergency authorization for people 18 and older in December.
“We are encouraged that the Moderna COVID-19 vaccine was highly effective at preventing COVID-19 and SARS-CoV-2 infection in adolescents,” Moderna CEO Stéphane Bancel said in a news release.
"We remain committed to helping to end the COVID-19 pandemic," he added.
Moderna also has applied for approval for its vaccine for adolescents in Canada and Europe and plans to file more requests in other countries, Bancel said.
If approved by the FDA, Moderna’s would be the second vaccine in the U.S. made available to adolescents. Pfizer’s vaccine received emergency use authorization for 12- to 15-year-olds last month — it had previously been approved for 16 and older.
The other COVID-19 vaccine authorized in the U.S., a single-shot dose from Johnson & Johnson, is available for ages 18 and older.